Remember, a long time has passed and a lot of people have died while Evidence-Based Medicine was facing the wrong way.
And asking the wrong question. "What new treatment will save more lives in the ICU?" is an important question, but one with few answers and no great ones - "What can stop people who catch SARS-CoV-2 coming to the ICU?" is a better one in a pandemic, and one that might also lead to better treatment protocols.
Selenium reduces COVID-19 mortality: A Bradford Hill analysis
Selenium reduces COVID-19 mortality: A Bradford Hill analysis
1) Strength of association. Very Strong.
a) On inspection of the Hubei data, it is notable that the cure rate in Enshi city, at 36.4%, was much higher than that of other Hubei cities, where the overall cure rate was 13.1% (Supplemental Table 1); indeed, the Enshi cure rate was significantly different from that in the rest of Hubei (P < 0.0001). Enshi is renowned for its high selenium intake and status [mean ± SD: hair selenium: 3.13 ± 1.91 mg/kg for females and 2.21 ± 1.14 mg/kg for males]—compare typical levels in Hubei of 0.55 mg/kg (10)—so much so that selenium toxicity was observed there in the 1960s. Selenium intake in Enshi was reported as 550 µg/d in 2013.
Similar inspection of data from provinces outside Hubei shows that Heilongjiang Province in northeast China, a notoriously low-selenium region in which Keshan is located, had a much higher death rate, at 2.4%, than that of other provinces (0.5%; P < 0.0001). The selenium intake was recorded as only 16 µg/d in a 2018 publication, while hair selenium in the Songnen Plain of Heilongjiang was measured as only 0.26 mg/kg (Supplemental Table 2).
Finally, we found a significant association between cure rate and background selenium status in cities outside Hubei (R2 = 0.72, F test P < 0.0001; Figure 1, Supplemental Table 2).[2]

b) Serum samples (n = 166) from COVID-19 patients (n = 33) were collected consecutively and analyzed for total Se by X-ray fluorescence and selenoprotein P (SELENOP) by a validated ELISA. Both biomarkers showed the expected strong correlation (r = 0.7758, p < 0.001), pointing to an insufficient Se availability for optimal selenoprotein expression. In comparison with reference data from a European cross-sectional analysis (EPIC, n = 1915), the patients showed a pronounced deficit in total serum Se (mean ± SD, 50.8 ± 15.7 vs. 84.4 ± 23.4 µg/L) and SELENOP (3.0 ± 1.4 vs. 4.3 ± 1.0 mg/L) concentrations. A Se status below the 2.5th percentile of the reference population, i.e., [Se] < 45.7 µg/L and [SELENOP] < 2.56 mg/L, was present in 43.4% and 39.2% of COVID samples, respectively.
The Se status was significantly higher in samples from surviving COVID patients as compared with non-survivors (Se; 53.3 ± 16.2 vs. 40.8 ± 8.1 µg/L, SELENOP; 3.3 ± 1.3 vs. 2.1 ± 0.9 mg/L), recovering with time in survivors while remaining low or even declining in non-survivors.[3]
c) Vitamins B1, B6, B12, D (25-hydroxyvitamin D), folate, selenium, and zinc levels were measured in 50 hospitalized patients with COVID-19. A total of 76% of the patients were vitamin D deficient and 42% were selenium deficient. No significant increase in the incidence of deficiency was found for vitamins B1, B6, and B12. folate, and zinc in patients with COVID-19. The COVID-19 group showed significantly lower vitamin D values than the healthy control group (150 people, age/sex matching). Severe vitamin D deficiency (based on 10 ng/dL) was found in 24% of the patients in the COVID-19 group and 7.3% of the control group. Among 12 patients with respiratory distress, 11 (91.7%) were deficient in at least one nutrient. However, patients without respiratory distress showed deficiency in 30/38 people (78.9%, P-value 0.425). These results suggest that a deficiency of vitamin D or selenium may decrease the immune defenses against COVID-19 and cause progression to severe disease; however, more precise and large-scale studies are needed.[18]
100% of the patients in this study with severe outcomes, including death, were selenium deficient; 75% were vitamin D deficient; none were zinc deficient.
d) In regression models, serum Se levels were inversely associated with lung damage independently of other markers of disease severity, anthropometric, biochemical, and hemostatic parameters.[23]
e) The association between soil Se level and the incidence of COVID-19 was observed in different cities of Hubei Province. The incidence of COVID-19 was more than 10 times lower in Se-enriched cities (Enshi, Shiyan, and Xiangyang) than in Se-deficient cities (Suizhou and Xiaogan).[25]
See also refs 19 and 22, discussed below.

b) Serum samples (n = 166) from COVID-19 patients (n = 33) were collected consecutively and analyzed for total Se by X-ray fluorescence and selenoprotein P (SELENOP) by a validated ELISA. Both biomarkers showed the expected strong correlation (r = 0.7758, p < 0.001), pointing to an insufficient Se availability for optimal selenoprotein expression. In comparison with reference data from a European cross-sectional analysis (EPIC, n = 1915), the patients showed a pronounced deficit in total serum Se (mean ± SD, 50.8 ± 15.7 vs. 84.4 ± 23.4 µg/L) and SELENOP (3.0 ± 1.4 vs. 4.3 ± 1.0 mg/L) concentrations. A Se status below the 2.5th percentile of the reference population, i.e., [Se] < 45.7 µg/L and [SELENOP] < 2.56 mg/L, was present in 43.4% and 39.2% of COVID samples, respectively.
The Se status was significantly higher in samples from surviving COVID patients as compared with non-survivors (Se; 53.3 ± 16.2 vs. 40.8 ± 8.1 µg/L, SELENOP; 3.3 ± 1.3 vs. 2.1 ± 0.9 mg/L), recovering with time in survivors while remaining low or even declining in non-survivors.[3]
c) Vitamins B1, B6, B12, D (25-hydroxyvitamin D), folate, selenium, and zinc levels were measured in 50 hospitalized patients with COVID-19. A total of 76% of the patients were vitamin D deficient and 42% were selenium deficient. No significant increase in the incidence of deficiency was found for vitamins B1, B6, and B12. folate, and zinc in patients with COVID-19. The COVID-19 group showed significantly lower vitamin D values than the healthy control group (150 people, age/sex matching). Severe vitamin D deficiency (based on 10 ng/dL) was found in 24% of the patients in the COVID-19 group and 7.3% of the control group. Among 12 patients with respiratory distress, 11 (91.7%) were deficient in at least one nutrient. However, patients without respiratory distress showed deficiency in 30/38 people (78.9%, P-value 0.425). These results suggest that a deficiency of vitamin D or selenium may decrease the immune defenses against COVID-19 and cause progression to severe disease; however, more precise and large-scale studies are needed.[18]
100% of the patients in this study with severe outcomes, including death, were selenium deficient; 75% were vitamin D deficient; none were zinc deficient.
d) In regression models, serum Se levels were inversely associated with lung damage independently of other markers of disease severity, anthropometric, biochemical, and hemostatic parameters.[23]
e) The association between soil Se level and the incidence of COVID-19 was observed in different cities of Hubei Province. The incidence of COVID-19 was more than 10 times lower in Se-enriched cities (Enshi, Shiyan, and Xiangyang) than in Se-deficient cities (Suizhou and Xiaogan).[25]
See also refs 19 and 22, discussed below.
2) Consistency - Very Strong
All epidemiological data about selenium and COVID-19 is consistent in direction and effect size. However, tests that could be done comparing COVID-19 risk in high and low selenium regions of Brazil, Scandinavia (selenium is supplemented in the food supply of Finland), and the USA would establish consistency further.
[edit 13/06/2022] - New study from the USA shows a two-fold higher mortality rate in low-selenium regions.[30]
Discussed in more detail on my Patreon blog - the only way I make any part of a living from this work, so help a brother out!
[edit 16/11/202o] - New study from South India is consistent with those from Germany, China, and South Korea:
We analysed the blood serum levels in apparently healthy (N=30) individuals and those with confirmed COVID -19 infection (N=30) in the southern part of India. Patients showed a significantly lower selenium level of 69.2 ±8.7 ng/ml than controls 79.1 ± 10.9 ng/ml, the difference was statistically significant (P=0.0003). Interestingly the controls showed a borderline level of selenium, suggesting that the level of this micronutrient is not optimum in the population studied.[19]
[edit 14/12/2020] letter from Finland in BJN compares death rate with Sweden's.
[edit 15/12/2020 deficiency of both zinc and selenium predicts COVID-19 severity in EPIC data]
"This combined deficit was observed in 0.15% of samples in the EPIC cohort of healthy subjects, in 19.7% of the samples collected from the surviving COVID-19 patients and in 50.0% of samples from the non-survivors."[22]
Statistically significant and often very strong associations between selenium intake, selenium status, and various COVID-19 outcomes have been reported from China, South Korea, Germany, South India, Russia and Europe. No null association has yet been reported.
Rigorous re-analysis of updated Chinese pandemic data published recently confirms the original observations, this time using the case-fatality rate:
A total of 147 cities each reporting over 20 cases were included in the current analysis. In these cities, 91% (14,045) of total cases and 85.8% (103) of total mortality from COVID-19 in China had been reported.
Totally, 14,045 COVID-19 cases were reported from 147 cities during 8 December 2019–13 December 2020 were included. Based on selenium content in crops, the case fatality rates (CFRs) gradually increased from 1.17% in non-selenium-deficient areas, to 1.28% in moderate-selenium-deficient areas, and further to 3.16% in severe-selenium-deficient areas (P = 0.002). Based on selenium content in topsoil, the CFRs gradually increased from 0.76% in non-selenium-deficient areas, to 1.70% in moderate-selenium-deficient areas, and further to 1.85% in severe-selenium-deficient areas (P < 0.001). The zero-inflated negative binomial regression model showed a significantly higher fatality risk in cities with severe-selenium-deficient selenium content in crops than non-selenium-deficient cities, with incidence rate ratio (IRR) of 3.88 (95% CIs: 1.21–12.52), which was further confirmed by regression fitting the association between CFR of COVID-19 and selenium content in topsoil, with the IRR of 2.38 (95% CIs: 1.14–4.98) for moderate-selenium-deficient cities and 3.06 (1.49–6.27) for severe-selenium-deficient cities.[24]
UPDATE 23/11/2021
A recent review of in-hospital selenium data shows consistent associations between lower Se and adverse outcomes in 9/10 comparisons where the population selenium level is below the optimal range of 130-150 mcg/dL. The outlier is an n=9 study (the smallest) in which length of hospital stay is the outcome and supplementation during the stay may be a confounder. In the one study where Se went over the optimal range a higher Se was found in more severe cases.[26]
3) Specificity - Strong
Selenium has much weaker or less consistent associations with other diseases, except those caused by other RNA viruses, e.g. when risk of hepatocellular cancer in viral hepatitis patients is compared with risk of osteoporosis.[4, 5]
4) Temporality - Strong
Prospective ecological comparisons are temporal by design.[2] In the German study, the temporal association between low serum selenium levels and COVID-19 symptom severity was closely tracked.[3]
5) Dose-response gradient - Very Strong
Selenium has much weaker or less consistent associations with other diseases, except those caused by other RNA viruses, e.g. when risk of hepatocellular cancer in viral hepatitis patients is compared with risk of osteoporosis.[4, 5]
4) Temporality - Strong
Prospective ecological comparisons are temporal by design.[2] In the German study, the temporal association between low serum selenium levels and COVID-19 symptom severity was closely tracked.[3]

5) Dose-response gradient - Very Strong
A strong, consistent dose-response is seen, even at levels where the risk of selenium toxicity exists, and despite the fact that toxic levels of soil selenium are often a legacy of industrial pollution in China.[2]
6) Plausibility - Very Strong
Reading references 2 and 3, as well as this review of the evidence written before reference 2 was published, should be persuasive.[6] See also ref 17 for antiviral effects. The effects of selenium and selenite align to support the associational results across multiple mechanisms.
7) Coherence - Very Strong
Selenium is well-studied and nothing in its story seems to contradict the idea that higher intakes will protect against COVID-19 mortality and reduce the severity of disease.
Dexamethasone, a drug which can reduce COVID-19 mortality in the ICU, enhances 1α,25-dihydroxyvitamin D3 effects by increasing vitamin D receptor transcription.[7]
Selenium sufficiency is essential for the function of vitamin D in peripheral blood monocytes.[8] Vitamin D status also correlates with COVID-19 survival.[9]
6) Plausibility - Very Strong
Reading references 2 and 3, as well as this review of the evidence written before reference 2 was published, should be persuasive.[6] See also ref 17 for antiviral effects. The effects of selenium and selenite align to support the associational results across multiple mechanisms.
7) Coherence - Very Strong
Selenium is well-studied and nothing in its story seems to contradict the idea that higher intakes will protect against COVID-19 mortality and reduce the severity of disease.
Dexamethasone, a drug which can reduce COVID-19 mortality in the ICU, enhances 1α,25-dihydroxyvitamin D3 effects by increasing vitamin D receptor transcription.[7]
Selenium sufficiency is essential for the function of vitamin D in peripheral blood monocytes.[8] Vitamin D status also correlates with COVID-19 survival.[9]
[Edit: 20/11/20] Two conditions which are associated with selenium depletion through effects on tubular mineral resorption, sickle cell disease (aOR, 1.73; 95% CI, 1.21-2.47), and chronic kidney disease (aOR, 1.32; 95% CI, 1.29-1.36), are the comorbidities most strongly associated with COVID-19 mortality in a large US MEDICARE patient analysis.[20] Selenium status in sickle cell disease is inversely associated with markers of hemolysis, a feature of severe COVID-19 pathology.[21]
8) Experiment - Weak (Neglected)
This is an area of sufficient neglect to make you despair about medical humanity, if you know that there have been thousands of trials of potentially useless drugs for COVID-19 already. However this criteria overlaps with the next section as there are several trials of selenium supplementation in other viral diseases, and animal experiments in analogous conditions, and many mechanistic experiments that are non-specific. The interaction between SARS-CoV-2 and selenoproteins has been confirmed by experiment.[10]
UPDATE 23/11/15
9) Analogy - Strong
Selenium intake is protective, and selenium supplementation has been useful, in other viral illnesses.
However, the protective effect of high selenium intakes before infection in epidemiology appears stronger than the protective effect of selenium as a late intervention in disease.[6, 11]
Those are the nine canonical Bradford Hill criteria. The discussion about selenium suggests that an ad hoc 10th criteria will also be useful:
This is an area of sufficient neglect to make you despair about medical humanity, if you know that there have been thousands of trials of potentially useless drugs for COVID-19 already. However this criteria overlaps with the next section as there are several trials of selenium supplementation in other viral diseases, and animal experiments in analogous conditions, and many mechanistic experiments that are non-specific. The interaction between SARS-CoV-2 and selenoproteins has been confirmed by experiment.[10]
UPDATE 23/11/15
This team in Wuerzburg Germany have ben steadily researching selenium in COVID-19 patients in ICU and have got to the stage of testing an intervention.
There's no control arm but we have proof of safety for 1mg sodium selenite and proof of concept in that people in whom the intervention raised SelenoP did better.
We don't yet know that this effect isn't an artifact of disease severity, but such careful work brings the needed RCT closer.
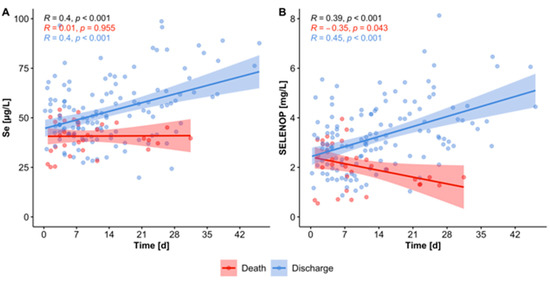
"According to intensive care unit (ICU) standard operating procedures, patients received 1.0 mg of intravenous Se daily on top of artificial nutrition, which contained various amounts of Se and Zn. Micronutrients, inflammatory cytokines, lymphocyte subsets and clinical data were extracted from the patient data management system on admission and after 10 to 14 days of treatment. Forty-six patients were screened for eligibility and 22 patients were included in the study. Twenty-one patients (95%) suffered from severe ARDS and 14 patients (64%) survived to ICU discharge. On admission, the majority of patients had low Se status biomarkers and Zn levels, along with elevated inflammatory parameters. Se supplementation significantly elevated Se (p = 0.027) and selenoprotein P levels (SELENOP; p = 0.016) to normal range. Accordingly, glutathione peroxidase 3 (GPx3) activity increased over time (p = 0.021). Se biomarkers, most notably SELENOP, were inversely correlated with CRP (rs = −0.495), PCT (rs = −0.413), IL-6 (rs = −0.429), IL-1β (rs = −0.440) and IL-10 (rs = −0.461). Positive associations were found for CD8+ T cells (rs = 0.636), NK cells (rs = 0.772), total IgG (rs = 0.493) and PaO2/FiO2 ratios (rs = 0.504). In addition, survivors tended to have higher Se levels after 10 to 14 days compared to non-survivors (p = 0.075). Sufficient Se and Zn levels may potentially be of clinical significance for an adequate immune response in critically ill patients with severe COVID-19 ARDS.[27]
In comparison to patients with a fatal outcome (n = 8), survivors (n = 14) significantly responded to supplementation with an increase in Se (p = 0.008), SELENOP (p = 0.004), GPx3 (p = 0.039) and Zn levels (p = 0.020) over the course of the ICU stay (Figure 5). Decedents had a median ICU course of 17.5 days (12–22), whereas patients with a favorable outcome were treated for significantly longer (40 days, 20–44; p = 0.025)."
There are also two tests of mixtures including selenium for COVID-19 with favourable results, the first is a survey of a clinic's patients already taking selenium, zinc, and vitamin D for Hasimoto's thyroiditis.[28]
After adjusting for age, gender, BMI, smoking status, we found an association between the absence of supplements and the risk of hospitalization, and invasive mechanical ventilation. Patients with Hashimoto’s thyroiditis who had COVID-19 infection and who had previously taken supplements such as selenium, zinc, and vitamin D had milder clinical outcomes, or no symptoms compared to those who did not receive supplements who had a moderate or severe outcome (P <0.05)
The next is an RCT from the South Indian doctors cited earlier, of a mixed supplement supplying 40 mcg selenium (a very modest dose in this context, but not insignificant), n=100.[29]
ImmuActiveTM 500 mg capsule containing curcuminoids (100 mg), andrographolides (50 mg), resveratrol (50 mg), zinc (10 mg), selenium (40 mcg), and piperine (3 mg) or placebo was administered orally to subjects once daily after breakfast.
Results. The ordinal scale at the end of the study was significantly lower in COVID-19 patients supplemented with ImmuActive (0.57) than placebo (1.0), with a value of 0.0043. The ordinal scale decreased by one unit within 2.35 days in ImmuActive-supplemented patients, while it took 3.36 days in placebo-supplemented patients. Days of hospitalization and time required to turn RT-PCR negative were comparatively lower in the ImmuActive arm than the placebo arm. Change in modified Jackson’s Symptom Severity Score and COVID-19 QOL were significant from screening to the end of the study in both ImmuActive and placebo arms. There were no adverse events observed during the study period.
There are also two tests of mixtures including selenium for COVID-19 with favourable results, the first is a survey of a clinic's patients already taking selenium, zinc, and vitamin D for Hasimoto's thyroiditis.[28]
After adjusting for age, gender, BMI, smoking status, we found an association between the absence of supplements and the risk of hospitalization, and invasive mechanical ventilation. Patients with Hashimoto’s thyroiditis who had COVID-19 infection and who had previously taken supplements such as selenium, zinc, and vitamin D had milder clinical outcomes, or no symptoms compared to those who did not receive supplements who had a moderate or severe outcome (P <0.05)
The next is an RCT from the South Indian doctors cited earlier, of a mixed supplement supplying 40 mcg selenium (a very modest dose in this context, but not insignificant), n=100.[29]
ImmuActiveTM 500 mg capsule containing curcuminoids (100 mg), andrographolides (50 mg), resveratrol (50 mg), zinc (10 mg), selenium (40 mcg), and piperine (3 mg) or placebo was administered orally to subjects once daily after breakfast.
Results. The ordinal scale at the end of the study was significantly lower in COVID-19 patients supplemented with ImmuActive (0.57) than placebo (1.0), with a value of 0.0043. The ordinal scale decreased by one unit within 2.35 days in ImmuActive-supplemented patients, while it took 3.36 days in placebo-supplemented patients. Days of hospitalization and time required to turn RT-PCR negative were comparatively lower in the ImmuActive arm than the placebo arm. Change in modified Jackson’s Symptom Severity Score and COVID-19 QOL were significant from screening to the end of the study in both ImmuActive and placebo arms. There were no adverse events observed during the study period.
9) Analogy - Strong
Selenium intake is protective, and selenium supplementation has been useful, in other viral illnesses.
However, the protective effect of high selenium intakes before infection in epidemiology appears stronger than the protective effect of selenium as a late intervention in disease.[6, 11]
Those are the nine canonical Bradford Hill criteria. The discussion about selenium suggests that an ad hoc 10th criteria will also be useful:
10) Risk - Weak in short-term, Well-Established in long term.
We can add the most relevant of extra questions to any given set of criteria - "strength of the alternative hypothesis" would be a good one for any lipid hypothesis.
Bradford Hill stated that some interventions are easier to justify than others.
[Edit: 1/09/20] There is also very good evidence that intravenous high dose selenite is safe in the ICU setting.
Totally 19 RCTs involving 3341 critically ill patients were carried out in which 1694 participates were in the selenium supplementation group, and 1647 in the control. The aggregated results suggested that compared with the control, intravenous selenium supplement as a single therapy could decrease the total mortality (RR = 0.86, 95% CI: 0.78–0.95, P = .002, TSA-adjusted 95% CI = 0.77–0.96, RIS = 4108, n = 3297) and may shorten the length of stay in hospital (MD −2.30, 95% CI −4.03 to −0.57, P = .009), but had no significant treatment effect on 28-days mortality (RR = 0.96, 95% CI: 0.85–1.09, P = .54) and could not shorten the length of ICU stay (MD −0.15, 95% CI −1.68 to 1.38, P = .84) in critically ill patients.[13]
This, and an earlier analysis which found less benefit, did not single out viral illnesses as a subgroup - this is only evidence for safety - but the earlier analysis did find a) slightly lower mortality in trials without an initial bolus dose, b) no increased risk in patients with renal disease.[14]
I will hypothesize briefly on selenium increasing mortality at 300 mcg/day in the Danish intervention study, a dose far too low to cause selenosis.
(The conventional signs of selenosis result from selenocysteine replacing cysteine in proteins, and the relative weakness of the Se-Se bond compared with the S-S bond.)
[Edit - hypothesis improved, 23/09/20]
It makes sense to me that selenomethionine, very useful as it will increase selenoprotein levels quickly if you don't have much time, should be replaced with sodium selenite for long-term coverage.
Brazil nuts are a variable quantity, a sample of nuts sold in NZ in 2008 had an average of 19 mcg per nut and increased selenoprotein levels more than selenomethionine.[16]
Plasma selenium increased by 64.2%, 61.0%, and 7.6%; plasma GPx by 8.3%, 3.4%, and -1.2%; and whole blood GPx by 13.2%, 5.3%, and 1.9% in the Brazil nut, selenomethionine, and placebo groups, respectively. Change over time at 12 wk in plasma selenium (P < 0.0001 for both groups) and plasma GPx activity in the Brazil nut (P < 0.001) and selenomethionine (P = 0.014) groups differed significantly from the placebo group but not from each other. The change in whole blood GPx activity was greater in the Brazil nut group than in the placebo (P = 0.002) and selenomethionine (P = 0.032) groups.
[Edit 02/09/20] - thanks to Mike Angell for this link; while all selenium sources are probably protective against death and ongoing harm from COVID-19, only selenite is likely to have an additional antiviral effect, and has low toxicity.[17]
A rational protocol for using selenium in prevention and treatment of COVID-19, fully consistent with the evidence discussed here, is described at the end of this paper:
https://www.frontiersin.org/articles/10.3389/fnut.2020.00164/full

We can add the most relevant of extra questions to any given set of criteria - "strength of the alternative hypothesis" would be a good one for any lipid hypothesis.
Bradford Hill stated that some interventions are easier to justify than others.
On fair evidence we might take action on what appears to be an occupational hazard, e.g. we might change from a probably carcinogenic oil to a non-carcinogenic oil in a limited environment and without too much injustice if we are wrong. But we should need very strong evidence before we made people burn a fuel in their homes that they do not like or stop smoking the cigarettes and eating the fats and sugar that they do like. In asking for very strong evidence I would, however, repeat emphatically that this does not imply crossing every ‘t’, and swords with every critic, before we act.[1]
With nutrient intakes there is often an identifiable risk, with a J-shaped curve. With selenium the risk is selenosis, which is a condition that requires chronic high exposure (I have given myself mild selenosis with around 900mcg selenium a day and it was not a terrible condition to experience and was reversible). There could be other risks. Luckily we have an experiment that tells us where the limit is.
In a low selenium country, like New Zealand or Denmark, you don't want to take more than 200mcg of extra selenium long term.[12] Pity the low dose arms here weren't retained in the intervention.

During 6871 person-years of follow-up, 158 deaths occurred. In an intention-to-treat analysis, the hazard ratio (95% confidence interval) for all-cause mortality comparing 300 µg selenium/d to placebo was 1.62 (0.66, 3.96) after 5 years of treatment and 1.59 (1.02, 2.46) over the entire follow-up period. The 100 and 200 µg/d doses showed non-significant decreases in mortality during the intervention period that disappeared after treatment cessation. Although we lacked power for endpoints other than all-cause mortality, the effects on cancer and cardiovascular mortality appeared similar.
Howsoever that may be, taking extra selenium above 200mcg per day may yet be advised if one becomes ill with COVID-19, but an inorganic salt of selenium like sodium selenite (which is anyhow probably safer than the selenomethionine form long-term, as I'll discuss below) is preferable, according to the selenovirus expert, Ethan Will Taylor.
(this video link does not show in the mobile version of this post but can be reached through the web view option at the bottom)
With nutrient intakes there is often an identifiable risk, with a J-shaped curve. With selenium the risk is selenosis, which is a condition that requires chronic high exposure (I have given myself mild selenosis with around 900mcg selenium a day and it was not a terrible condition to experience and was reversible). There could be other risks. Luckily we have an experiment that tells us where the limit is.
In a low selenium country, like New Zealand or Denmark, you don't want to take more than 200mcg of extra selenium long term.[12] Pity the low dose arms here weren't retained in the intervention.

During 6871 person-years of follow-up, 158 deaths occurred. In an intention-to-treat analysis, the hazard ratio (95% confidence interval) for all-cause mortality comparing 300 µg selenium/d to placebo was 1.62 (0.66, 3.96) after 5 years of treatment and 1.59 (1.02, 2.46) over the entire follow-up period. The 100 and 200 µg/d doses showed non-significant decreases in mortality during the intervention period that disappeared after treatment cessation. Although we lacked power for endpoints other than all-cause mortality, the effects on cancer and cardiovascular mortality appeared similar.
Howsoever that may be, taking extra selenium above 200mcg per day may yet be advised if one becomes ill with COVID-19, but an inorganic salt of selenium like sodium selenite (which is anyhow probably safer than the selenomethionine form long-term, as I'll discuss below) is preferable, according to the selenovirus expert, Ethan Will Taylor.
(this video link does not show in the mobile version of this post but can be reached through the web view option at the bottom)
[Edit: 1/09/20] There is also very good evidence that intravenous high dose selenite is safe in the ICU setting.
Totally 19 RCTs involving 3341 critically ill patients were carried out in which 1694 participates were in the selenium supplementation group, and 1647 in the control. The aggregated results suggested that compared with the control, intravenous selenium supplement as a single therapy could decrease the total mortality (RR = 0.86, 95% CI: 0.78–0.95, P = .002, TSA-adjusted 95% CI = 0.77–0.96, RIS = 4108, n = 3297) and may shorten the length of stay in hospital (MD −2.30, 95% CI −4.03 to −0.57, P = .009), but had no significant treatment effect on 28-days mortality (RR = 0.96, 95% CI: 0.85–1.09, P = .54) and could not shorten the length of ICU stay (MD −0.15, 95% CI −1.68 to 1.38, P = .84) in critically ill patients.[13]
This, and an earlier analysis which found less benefit, did not single out viral illnesses as a subgroup - this is only evidence for safety - but the earlier analysis did find a) slightly lower mortality in trials without an initial bolus dose, b) no increased risk in patients with renal disease.[14]
I will hypothesize briefly on selenium increasing mortality at 300 mcg/day in the Danish intervention study, a dose far too low to cause selenosis.
(The conventional signs of selenosis result from selenocysteine replacing cysteine in proteins, and the relative weakness of the Se-Se bond compared with the S-S bond.)
[Edit - hypothesis improved, 23/09/20]
The question of selenium causing insulin resistance and increasing mortality in high-dose supplements, not mirrored as far as I can see in natural high-dose populations, may have a simple explanation - supplements allow us to consume micronutrients without protein.
If you have no cysteine or methionine coming in when you take Se (either because you're not eating protein, or perhaps it can happen naturally if the Se level is high in a low-protein food and diet) then the selenocysteine formed will be incorporated into all proteins, not just the ones that require it. Including the insulin receptors, which will suffer a relative loss of function.
(similarly, though for different reasons, pyridoxine toxicity can be triggered by supplementing on a low-protein diet)
If we think that insulin resistance causes CVD, then the increased risk from (mostly) natural high Se levels is not great, see fig 5 here [15], but the intervention studies have more alarming results, and I think the competition of selenium- vs sulphur-amino acids in protein fed vs unfed states can explain this. There is next to no evidence of Se toxicity from Brazil nuts, which are high in both Se and protein.
Brazil nuts are a variable quantity, a sample of nuts sold in NZ in 2008 had an average of 19 mcg per nut and increased selenoprotein levels more than selenomethionine.[16]
Plasma selenium increased by 64.2%, 61.0%, and 7.6%; plasma GPx by 8.3%, 3.4%, and -1.2%; and whole blood GPx by 13.2%, 5.3%, and 1.9% in the Brazil nut, selenomethionine, and placebo groups, respectively. Change over time at 12 wk in plasma selenium (P < 0.0001 for both groups) and plasma GPx activity in the Brazil nut (P < 0.001) and selenomethionine (P = 0.014) groups differed significantly from the placebo group but not from each other. The change in whole blood GPx activity was greater in the Brazil nut group than in the placebo (P = 0.002) and selenomethionine (P = 0.032) groups.
[Edit 02/09/20] - thanks to Mike Angell for this link; while all selenium sources are probably protective against death and ongoing harm from COVID-19, only selenite is likely to have an additional antiviral effect, and has low toxicity.[17]
A rational protocol for using selenium in prevention and treatment of COVID-19, fully consistent with the evidence discussed here, is described at the end of this paper:
https://www.frontiersin.org/articles/10.3389/fnut.2020.00164/full

All scientific work is incomplete - whether it be observational or experimental. All scientific work is liable to be upset or modified by advancing knowledge. That does not confer upon us a freedom to ignore the knowledge we already have, or to postpone the action that it appears to demand at a given time.
Austin Bradford Hill, 1965.
References:
[1] Hill AB. The environment and disease: association or causation? Proc R Soc Med. 1965;58(5):295-300.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1898525/pdf/procrsmed00196-0010.pdf[2] Jinsong Zhang, Ethan Will Taylor, Kate Bennett, Ramy Saad, Margaret P Rayman, Association between regional selenium status and reported outcome of COVID-19 cases in China, The American Journal of Clinical Nutrition, Volume 111, Issue 6, June 2020, Pages 1297–1299, https://doi.org/10.1093/ajcn/nqaa095
[3] Moghaddam, A.; Heller, R.A.; Sun, Q.; Seelig, J.; Cherkezov, A.; Seibert, L.; Hackler, J.; Seemann, P.; Diegmann, J.; Pilz, M.; Bachmann, M.; Minich, W.B.; Schomburg, L. Selenium Deficiency Is Associated with Mortality Risk from COVID-19. Nutrients 2020, 12, 2098.
[4] Yu MW, Horng IS, Hsu KH, Chiang YC, Liaw YF, Chen CJ. Plasma selenium levels and risk of hepatocellular carcinoma among men with chronic hepatitis virus infection. Am J Epidemiol. 1999;150(4):367-374. doi:10.1093/oxfordjournals.aje.a010016
[4] Yu MW, Horng IS, Hsu KH, Chiang YC, Liaw YF, Chen CJ. Plasma selenium levels and risk of hepatocellular carcinoma among men with chronic hepatitis virus infection. Am J Epidemiol. 1999;150(4):367-374. doi:10.1093/oxfordjournals.aje.a010016
[5] Wang, Y., Xie, D., Li, J. et al. Association between dietary selenium intake and the prevalence of osteoporosis: a cross-sectional study. BMC Musculoskelet Disord 20, 585 (2019). https://doi.org/10.1186/s12891-019-2958-5
[6] Bermano, G., Méplan, C., Mercer, D., & Hesketh, J. (2020). Selenium and viral infection: Are there lessons for COVID-19? British Journal of Nutrition, 1-37. doi:10.1017/S0007114520003128
https://www.cambridge.org/core/journals/british-journal-of-nutrition/article/selenium-and-viral-infection-are-there-lessons-for-covid19/BE3AC78D5C92725BE83C4E474ECBB548
[7] Hidalgo AA, Deeb KK, Pike JW, Johnson CS, Trump DL. Dexamethasone enhances 1alpha,25-dihydroxyvitamin D3 effects by increasing vitamin D receptor transcription. J Biol Chem. 2011;286(42):36228-36237. doi:10.1074/jbc.M111.244061
[8] Schütze N, Fritsche J, Ebert-Dümig R, et al. The selenoprotein thioredoxin reductase is expressed in peripheral blood monocytes and THP1 human myeloid leukemia cells--regulation by 1,25-dihydroxyvitamin D3 and selenite. Biofactors. 1999;10(4):329-338. doi:10.1002/biof.5520100403
[9] Martín Giménez, V.M., Inserra, F., Ferder, L. et al. Vitamin D deficiency in African Americans is associated with a high risk of severe disease and mortality by SARS-CoV-2. J Hum Hypertens (2020). https://doi.org/10.1038/s41371-020-00398-z
[10] Wang, Y et al. SARS-CoV-2 suppresses mRNA expression of selenoproteins associated with ferroptosis, ER stress and DNA synthesis. Preprint, 2020/07/31. 10.1101/2020.07.31.230243
https://www.researchgate.net/publication/343365020_SARS-CoV-2_suppresses_mRNA_expression_of_selenoproteins_associated_with_ferroptosis_ER_stress_and_DNA_synthesis[11] Steinbrenner H, Al-Quraishy S, Dkhil MA, Wunderlich F, Sies H. Dietary selenium in adjuvant therapy of viral and bacterial infections. Adv Nutr. 2015;6(1):73-82. Published 2015 Jan 15. doi:10.3945/an.114.007575
[12] Rayman MP, Winther KH, Pastor-Barriuso R, et al. Effect of long-term selenium supplementation on mortality: Results from a multiple-dose, randomised controlled trial. Free Radic Biol Med. 2018;127:46-54. doi:10.1016/j.freeradbiomed.2018.02.015
[13] Zhao Y, Yang M, Mao Z, et al. The clinical outcomes of selenium supplementation on critically ill patients: A meta-analysis of randomized controlled trials. Medicine (Baltimore). 2019;98(20):e15473. doi:10.1097/MD.0000000000015473
[14] Manzanares W, Lemieux M, Elke G, Langlois PL, Bloos F, Heyland DK. High-dose intravenous selenium does not improve clinical outcomes in the critically ill: a systematic review and meta-analysis. Crit Care. 2016;20(1):356. Published 2016 Oct 28. doi:10.1186/s13054-016-1529-5
[15] Angelica Kuria, Hongdou Tian, Mei Li, Yinhe Wang, Jan Olav Aaseth, Jiajie Zang & Yang Cao (2020) Selenium status in the body and cardiovascular disease: a systematic review and meta-analysis, Critical Reviews in Food Science and Nutrition, DOI: 10.1080/10408398.2020.1803200 https://www.tandfonline.com/doi/full/10.1080/10408398.2020.1803200
[16] Thomson CD, Chisholm A, McLachlan SK, Campbell JM. Brazil nuts: an effective way to improve selenium status. Am J Clin Nutr. 2008;87(2):379-384. doi:10.1093/ajcn/87.2.379
https://academic.oup.com/ajcn/article/87/2/379/4633360
[17] Kieliszek M, Lipinski B. Selenium supplementation in the prevention of coronavirus infections (COVID-19) [published online ahead of print, 2020 May 24]. Med Hypotheses. 2020;143:109878. doi:10.1016/j.mehy.2020.109878
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7246001/
[18] Im, JH et al. Nutritional status of patients with coronavirus disease 2019 (COVID-19) Int J Infectious Diseases, August 11, 2020
https://www.ijidonline.com/article/S1201-9712(20)30647-0/fulltext
https://www.ijidonline.com/article/S1201-9712(20)30647-0/fulltext
[19] Majeed, M et al. An Exploratory Study of Selenium Status in Normal Subjects and COVID-19 Patients in South Indian population: Case for Adequate Selenium Status: Selenium Status in COVID-19 Patients. Nutrition. Available online 11 November 2020, 111053
https://www.sciencedirect.com/science/article/pii/S0899900720303361
[20] Chen Dun, Christi M. Walsh, Sunjae Bae, Amesh Adalja, Eric Toner, Timothy A. Lash, Farah Hashim, Joseph Paturzo, Dorry L. Segev, Martin A. Makary. A Machine Learning Study of 534,023 Medicare Beneficiaries with COVID-19: Implications for Personalized Risk Prediction.
https://www.sciencedirect.com/science/article/pii/S0899900720303361
[20] Chen Dun, Christi M. Walsh, Sunjae Bae, Amesh Adalja, Eric Toner, Timothy A. Lash, Farah Hashim, Joseph Paturzo, Dorry L. Segev, Martin A. Makary. A Machine Learning Study of 534,023 Medicare Beneficiaries with COVID-19: Implications for Personalized Risk Prediction.
medRxiv 2020.10.27.20220970; doi: https://doi.org/10.1101/2020.10.27.20220970
[21] Delesderrier E, Cople-Rodrigues CS, Omena J, et al. Selenium Status and Hemolysis in Sickle Cell Disease Patients. Nutrients. 2019;11(9):2211. Published 2019 Sep 13. doi:10.3390/nu11092211
[22] Raban Arved Heller, Qian Sun, Julian Hackler, Julian Seelig, Linda Seibert, Asan Cherkezov, Waldemar B. Minich, Petra Seemann, Joachim Diegmann, Maximilian Pilz, Manuel Bachmann, Alireza Ranjbar, Arash Moghaddam, Lutz Schomburg,
Prediction of survival odds in COVID-19 by zinc, age and selenoprotein P as composite biomarker, Redox Biology, Volume 38, 2021, 101764,
[23] Skalny AV, Timashev PS, Aschner M, et al. Serum Zinc, Copper, and Other Biometals Are Associated with COVID-19 Severity Markers. Metabolites. 2021;11(4):244. Published 2021 Apr 15. doi:10.3390/metabo11040244
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8071197/
[24] Zhang, HY., Zhang, AR., Lu, QB. et al. Association between fatality rate of COVID-19 and selenium deficiency in China. BMC Infect Dis 21, 452 (2021). https://doi.org/10.1186/s12879-021-06167-8
[25] Liu Q, Zhao X, Ma J, et al. Selenium (Se) plays a key role in the biological effects of some viruses: Implications for COVID-19. Environ Res. 2021;196:110984. doi:10.1016/j.envres.2021.110984
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7937041/
[26] Fakhrolmobasheri, M., Mazaheri-Tehrani, S., Kieliszek, M. et al. COVID-19 and Selenium Deficiency: a Systematic Review. Biol Trace Elem Res (2021). https://doi.org/10.1007/s12011-021-02997-4
https://link.springer.com/article/10.1007/s12011-021-02997-4
[27] Notz, Q.; Herrmann, J.; Schlesinger, T.; Helmer, P.; Sudowe, S.; Sun, Q.; Hackler, J.; Roeder, D.; Lotz, C.; Meybohm, P.; Kranke, P.; Schomburg, L.; Stoppe, C. Clinical Significance of Micronutrient Supplementation in Critically Ill COVID-19 Patients with Severe ARDS. Nutrients 2021, 13, 2113.
https://doi.org/10.3390/nu13062113
[28] Zelija Velija Asimi, Almira Hadzovic-Dzuvo, & Djinan Al Tawil. Selenium, zinc, and vitamin D supplementation affect the clinical course of COVID-19 infection in Hashimoto’s thyroiditis. Presented ePosters 14: COVID-19 Endocrine Abstracts (2021) 73 PEP14.2 | DOI: 10.1530/endoabs.73.PEP14.2
https://www.endocrine-abstracts.org/ea/0073/ea0073pep14.2
[29] Muhammed Majeed, Kalyanam Nagabhushanam, Kalpesh Shah, Lakshmi Mundkur, "A Randomized, Double-Blind, Placebo-Controlled Study to Assess the Efficacy and Safety of a Nutritional Supplement (ImmuActiveTM) for COVID-19 Patients", Evidence-Based Complementary and Alternative Medicine, vol. 2021, Article ID 8447545, 9 pages, 2021. https://doi.org/10.1155/2021/8447545
[30] JinsongZhanga, EthanWill Taylorb, KateBennett, Margaret P. Rayman. Does atmospheric dimethyldiselenide play a role in reducing COVID-19 mortality?
Gondwana Research, Available online 6 June 2022 https://doi.org/10.1016/j.gr.2022.05.017[29] Muhammed Majeed, Kalyanam Nagabhushanam, Kalpesh Shah, Lakshmi Mundkur, "A Randomized, Double-Blind, Placebo-Controlled Study to Assess the Efficacy and Safety of a Nutritional Supplement (ImmuActiveTM) for COVID-19 Patients", Evidence-Based Complementary and Alternative Medicine, vol. 2021, Article ID 8447545, 9 pages, 2021. https://doi.org/10.1155/2021/8447545
[30] JinsongZhanga, EthanWill Taylorb, KateBennett, Margaret P. Rayman. Does atmospheric dimethyldiselenide play a role in reducing COVID-19 mortality?



4 comments:
George,
As you know histamine facilitates the entry of Covid into cells. Selenium and zinc inhibit mast cell release and decrease histamine.
https://link.springer.com/article/10.1007/s12011-013-9712-x?fbclid=IwAR3iGfQrTpdYAV8Vsi9-Zr_coD-4VGHdHK-CL38PpDZpUfqUGSU86KH7yv0
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6286780/
In ICU Fentanyl and Propofol IV Are used to keep vent patients sedated. Both release histamine in the lungs.
Of course, this is anecdotal, but my husband and my sister, both of whom live with me and are my caregivers (I had a stroke 10 years ago and am in a wheelchair) have had Covid in the last month (my husband is still recovering, both were mild cases, all of us were vaccinated but not boosted). I have been wondering why I haven't gotten it, but they did. When I read this today I realized this may be part of the answer. I've taken a minimal (20 mcg.) dose of selenium for decades, as I had read about it in cancer research back then (my mother died of cancer). Neither of them does. I also supplement Vitamin D, zinc, and Vitamin C, so confounders. But I guess I'll keep taking selenium.
Interesting Silvia - all opioids dump histamine, this is why you itch, and lower it in the brain - could be protective.
Post a Comment